Project Overview
EROSTER is an enterprise-grade event management platform built for pharmaceutical organizations to plan, govern, and audit medical events across regions, teams, and stakeholders.
The problem was not the absence of tools, but the absence of orchestration. Event operations relied on fragmented systems, manual coordination, and human follow-ups—creating operational risk, compliance gaps, and poor visibility.
Inspired by automation-led enterprise platforms, this initiative focused on reducing manual effort through structured workflows while maintaining human control, compliance, and accountability.
Problem
Pharmaceutical organizations lacked a unified, professional system to plan and manage medical events across teams, locations, and stakeholders.
- Fragmented workflows across planning, registration, communication, and reporting
- Manual coordination between internal teams, hosts, and participants
- Limited visibility into event progress, confirmations, and attendance
- Inconsistent standards for RSVP, access control, and notifications
- No centralized system for tracking events with auditability
Role & Ownership
- Lead UX Designer — concept to implementation
- User research, ideation, prototyping, and usability testing
- High-fidelity UI design and design standards definition
- Close collaboration with product, engineering, and stakeholders
- Exploration of emerging technologies including ML concepts
Strategy
- Design a role-based, end-to-end event management platform
- Establish a consistent UX and visual language across devices and roles
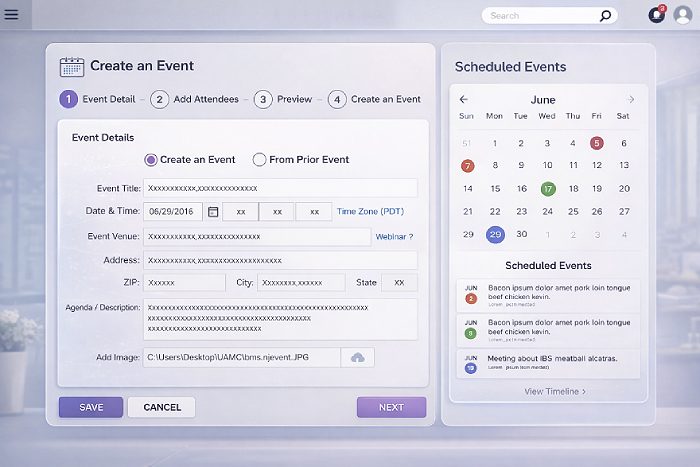
- Simplify complex workflows using guided event creation and task-based flows
- Introduce standardized, reusable modules for core event operations
- Ensure scalability through design standards and reusable UI patterns
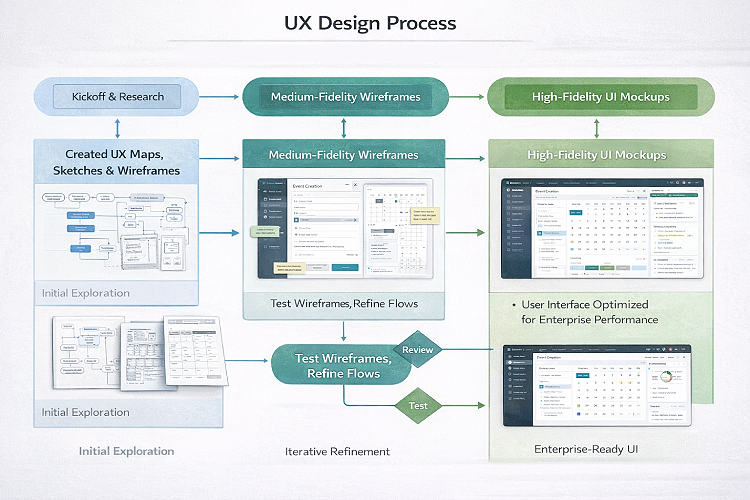
Execution
Research-driven design anchored execution. User interviews, journey mapping, and iterative validation shaped workflows centered on the Event Manager role.

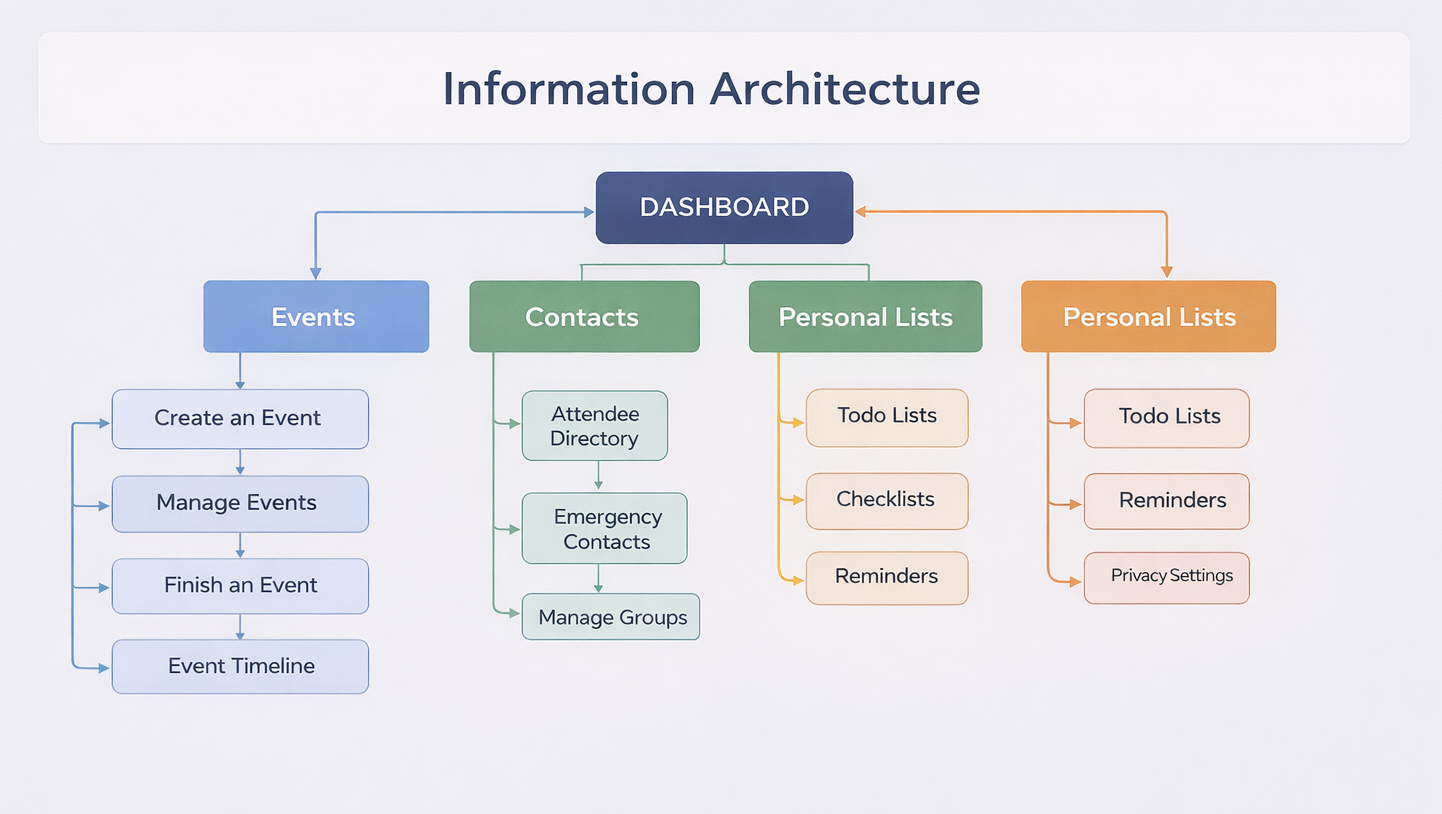
INFORMATION ARCHITECTURE
Pharmaceutical Event Management Platform (Web, Tablet, Mobile)
The information architecture is designed to support regulated medical events with a focus on clarity, compliance, and real-time visibility across devices. It enables structured execution while reducing manual coordination and operational risk.
The information architecture is designed to support regulated pharmaceutical events while staying practical for everyday use across web, tablet, and mobile. At the center is the dashboard, which acts as a single source of truth—giving teams a clear view of event status, pending actions, approvals, and critical alerts so nothing relies on memory or follow-ups.
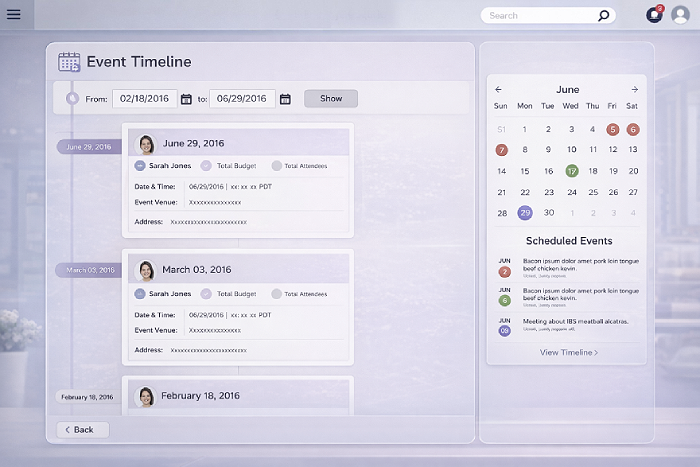
Events are organized around the full lifecycle, from creation through execution to closure. This structure makes it easy to manage tasks, RSVPs, attendance, and documentation while automatically building an audit-ready trail through the event timeline. Every action is visible, traceable, and tied to ownership, reducing operational risk without adding complexity.
Contacts and personal tools support the human side of event execution. Stakeholders, HCPs, and teams are managed in one place, with quick access to emergency information when events are live. To-do lists, checklists, and reminders help users stay on track without feeling overwhelmed. Personal settings give individuals control over notifications and data visibility, which is essential in a regulated environment. Overall, the architecture mirrors how people actually run medical events—clear, role-driven, and dependable—so teams can focus on execution with confidence rather than managing the system itself.
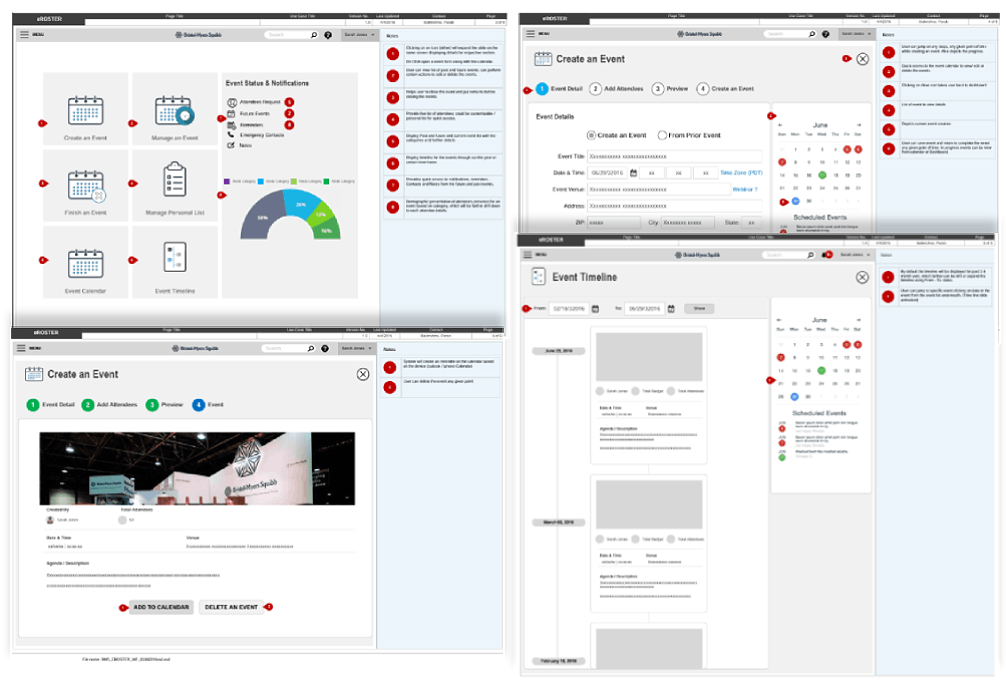
Wireframes
Impact
- Enabled end-to-end visibility across the event lifecycle
- Reduced manual coordination through standardized workflows
- Improved organizer efficiency and operational confidence
Final UI